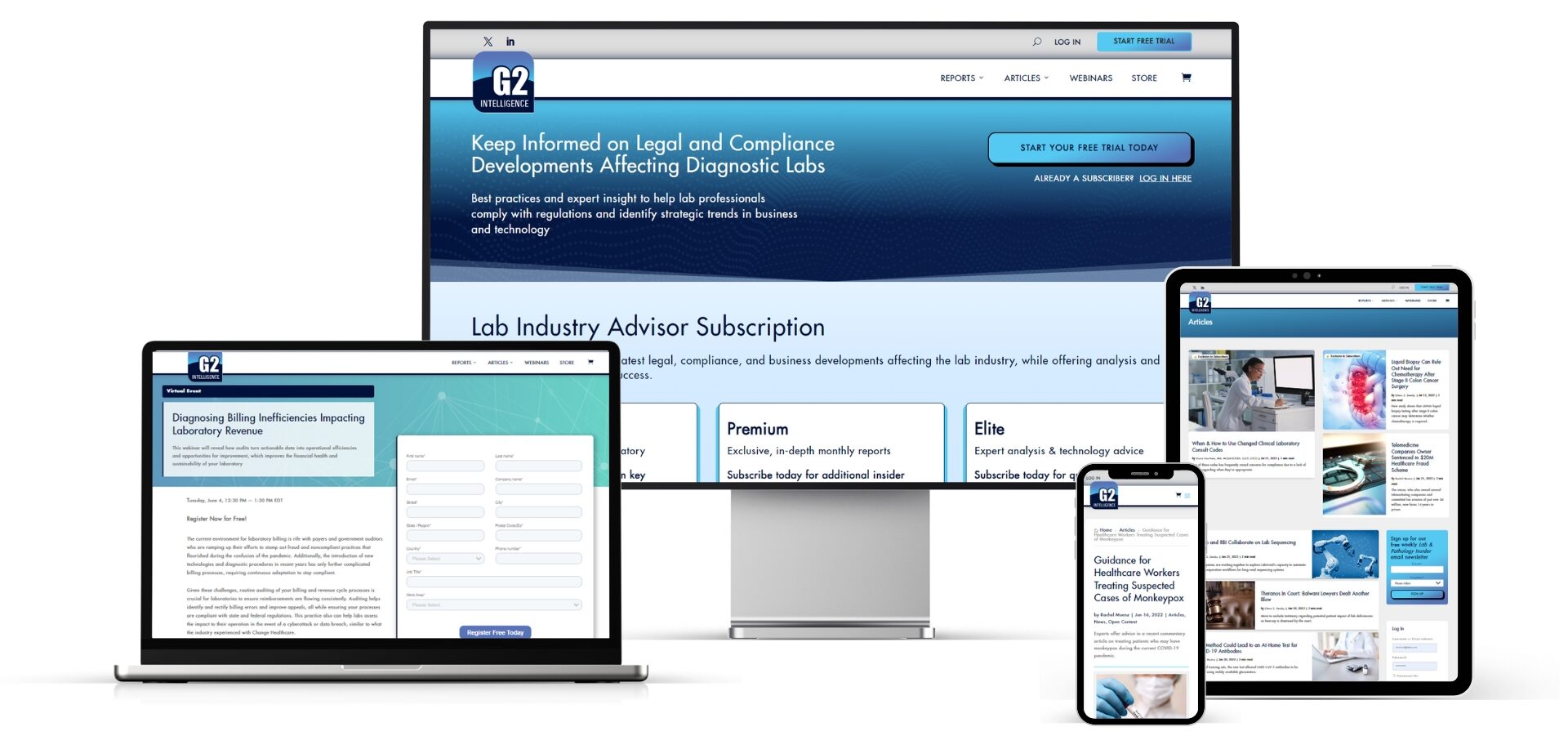
Founded over 30 years ago, G2 Intelligence is a premier information services provider catering to the dynamic needs of the medical laboratory industry. Our mission is to empower lab managers, professionals, and industry leaders to navigate legal requirements, optimize operational efficiency, enhance clinical quality, and drive profitability.
Monthly Reports

Lab Industry Advisor is a subscription-based, online briefing that keeps readers up to date on the latest legal, compliance, and business developments affecting the lab industry. Lab Industry Advisor offers analysis and insight from expert sources to help lab leaders meet their financial and business goals.

Clinical Diagnostics Insider, a spinoff of Lab Industry Advisor, is a subscription-based, online resource aimed at clinical lab scientists. It provides in-depth expert analysis of emerging diagnostics tests and disruptive technologies in the lab industry.
Webinars & Digital Events
Webinar presentations offer expert advice and education on critical topics for medical lab professionals, covering the legal, regulatory, compliance, and science and technology aspects of the industry throughout the year. Our 2024 calendar includes 11 webinars.

Lab Institute is a free virtual, two-day event including a variety of experts focused on one key theme in the medical lab industry. Including at least six presentations, our virtual events help lab leaders stay current on the latest requirements for labs and connect them with experts who offer solutions to the most pressing problems facing medical labs today. We plan to offer both a spring and fall Lab Institute in 2024.











