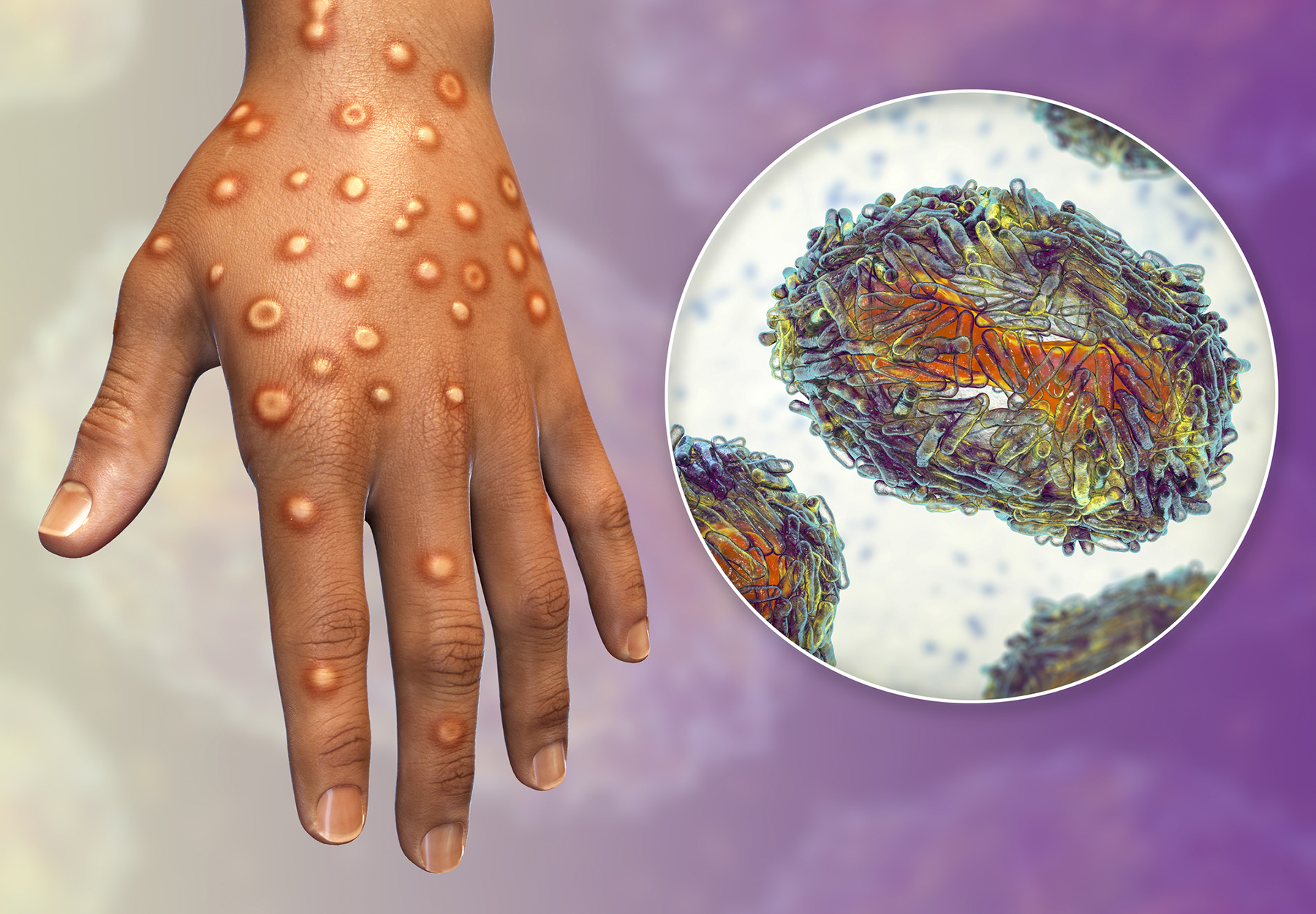
Initially characterized by the World Health Organization (WHO) as a “moderate” concern, the monkeypox outbreak is growing so fast that the agency declared it a “public health emergency of international concern” on July 23. While Europe has been most affected by the current outbreak, public health agencies are urging all countries to be on high alert as this form of the virus has symptoms different from typical monkeypox cases.
Meanwhile, the laboratories, health centers, and commercial test manufacturers that responded heroically to the COVID-19 pandemic by developing new tests for the SARS-CoV-2 virus in record time are now seeking to do the same for monkeypox. Here is a briefing on the current state of monkeypox products in the US.
The CDC Test & LRN Response Effort
As it did with COVID-19, the US Centers for Disease Control and Prevention (CDC) has developed a molecular real-time polymerase chain reaction (RT-PCR) test for monkeypox that it is currently distributing for free to laboratories across the country via its Laboratory Response Network (LRN). On June 22, the CDC began shipping the orthopoxvirus test, which detects most non-smallpox related orthopoxviruses, including monkeypox, to five commercial laboratory companies, including the nation’s largest reference laboratories, to quickly increase monkeypox testing capacity and access. The companies include Aegis Sciences, Labcorp, Mayo Clinic Laboratories, Quest Diagnostics, and Sonic Healthcare.
On July 6, Labcorp became the first national laboratory to offer the CDC test out of its largest US laboratory. According to the company, the plan is to expand testing from that North Carolina location to others on an as-needed basis. The laboratory has the capacity to take samples from its customers as well as any overflow from public health labs, but plans to increase capacity to a maximum of 10,000 tests each week, thereby doubling the current capacity provided through the LRN.
Mayo Clinic Laboratories, which became the second commercial laboratory to provide the CDC test on July 11, says that it can accept specimens from anywhere in the country and that it expects to be able to perform up to 10,000 tests per week. Aegis Sciences and Sonic Healthcare became the third and fourth commercial labs to offer the CDC test on July 14 and July 18, respectively, with Aegis offering a capacity of 10,000 tests per week and Sonic Healthcare expanding its capacity as needed, according to the CDC. As of July 18, the combined testing capacity of the CDC’s LRN and the five commercial labs was 80,000 specimens per week, up from an initial capacity of only 6,000.
LDTs for Monkeypox
As with the COVID-19 response, laboratories and test makers have bolstered the nationwide test response effort by generating their own laboratory developed tests (LDTs).
Roche Diagnostics
Only a few weeks after monkeypox cases began breaking out in Europe, Swiss firm Roche Diagnostics developed tests for the virus. This is hardly surprising, given that Roche’s RT PCR test was one of the first tests to receive Emergency Use Authorization (EUA) from the FDA in the early days of the COVID-19 crisis. With the help of subsidiary TIB Molbiol, Roche has launched three different LightMix Modular Virus kits for monkeypox:
- The first test detects orthopoxviruses, the biological grouping which includes viruses associated with monkeypox as well as smallpox and cowpox;
- The second test detects only monkeypox viruses, specifically the West African and Central African strains; and
- The third is for researchers and detects both orthopoxviruses and the monkeypox virus.
Bioperfectus
On May 23, Chinese firm Bioperfectus announced the launch of its Monkeypox Virus Real Time PCR Kit, a CE-marked assay for detecting the F3L gene areas of the virus. The kit also comes with a housekeeping gene (RNase P) as an internal control (IC) for specimen sampling and nucleic acid extraction.
Co-Diagnostics
On May 26, molecular diagnostics firm Co-Diagnostics announced that it had completed the principal design work for a PCR-based monkeypox test based on its CoPrimer technology and software system. Investors loved what they heard, fueling a seven percent increase in Co-Diagnostics stock on a day that the entire S&P 500 index gained only two percent.
Becton Dickinson-CerTest Collaboration
In the middle of June, Becton Dickinson (BD) and Spanish molecular test maker CerTest Biotec announced a collaboration to develop a PCR-based test for monkeypox to run on the BD Max System. The plan calls for using BD Max open system reagents to validate CerTest’s CE-IVD-marked Viasure Monkeypox molecular test. The firms will offer the resulting Monkeypox PCR Detection Kit for the BD Max in a lyophilized format that comes in a tube and snaps into the BD Max ExK TNA extraction kit.
Gold Standard Diagnostics
On June 23, Eurofins company Gold Standard Diagnostics announced the launch of Mplex Monkeypox, Orthopox, a multiplex RT PCR assay that can detect monkeypox and also differentiate between monkeypox and other orthopoxviruses.
The Cepheid-BioGX Collaboration
On June 24, Cepheid and BioGX announced a collaboration to create a PCR assay for monkeypox on the former’s GeneXpert, a system with an installed base of over 40,000 instruments in 180 countries. The assay will be provided as part of Cepheid’s FleXible Cartridge for GeneXpert, the companies stated.
Novacyt
On June 28, Novacyt announced the launch of its RUO PCR monkeypox assay that is available in two forms, genesig easy and genesig advanced, to detect both West African and Central African variants of the monkeypox virus genome.
Anitoa Systems
On June 29, Anitoa Systems joined the market by launching its CE-marked, “turn-key” PCR solution, including a real-time PCR instrument and extraction-free multiplex PCR reagent to test skin lesion samples from patients suspected of having monkeypox. The test can be carried out at the point of care.
Neelyx Labs
On July 11, independent diagnostics firm Neelyx Labs announced it is now processing monkeypox tests, with a capacity of 8,000 tests each day. The firm has released a CDC-compliant PCR test for monkeypox that allows for testing saliva, lesions, swabs in transport media (wet swabs), and dry swabs, the company stated.
Quest Diagnostics
On July 13, Quest Diagnostics, which plans to begin offering the CDC test in August, launched its own laboratory-developed molecular diagnostic test for monkeypox, a dual RT PCR test for qualitative detection and differentiation of monkeypox virus (West African clade) DNA from other non-variola orthopoxviruses. With the new automated test, Quest says it expects to be able to perform roughly 30,000 monkeypox virus tests a week by the end of July and that it can expand capacity for monkeypox testing across other advanced laboratories in its network if demand dictates.
Twist Bioscience
On July 14, high-quality synthetic DNA producer Twist Bioscience launched two human virus synthetic DNA controls for use in developing, verifying, and ongoing validation of both PCR and next-generation sequencing assays for monkeypox, covering over 80 percent of the full viral genome.
Editor’s Note: This article was updated on July 25, 2022, to reflect the WHO’s declaration that the monkeypox outbreak now represents a public health emergency of international concern. It was also updated on July 27, to include updated information on monkeypox testing offered through the five large commercial labs working in partnership with the CDC.