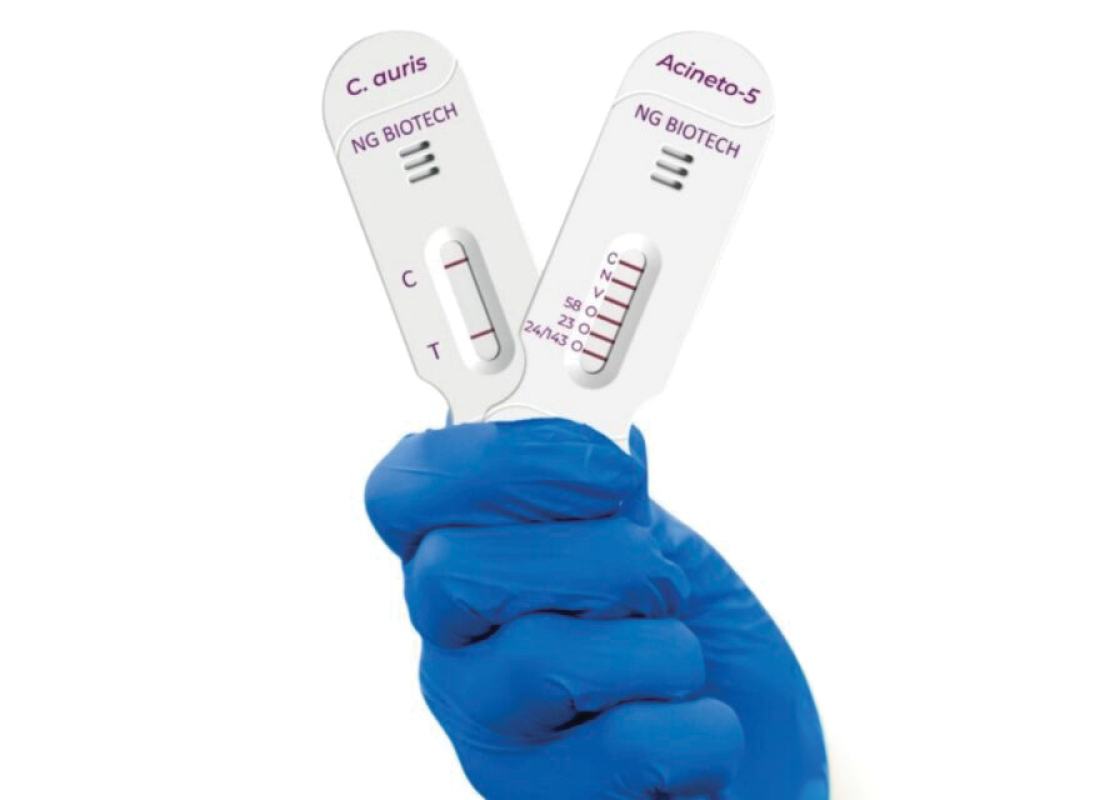
FDA Watch: MeMed and NG Biotech Receive FDA Breakthrough Device Designations for Rapid Infection Tests
In both cases, the diagnostic tests report results back within 15 minutes
Subscribe to view Essential
Start a Free Trial for immediate access to this article