
FDA Watch: New RAPID Coverage Pathway Would Enable Some Lab Tests to Pursue Clearance and Medicare Coverage Simultaneously
FDA-CMS collaboration aims to accelerate Medicare coverage determinations of certain medical devices

FDA-CMS collaboration aims to accelerate Medicare coverage determinations of certain medical devices
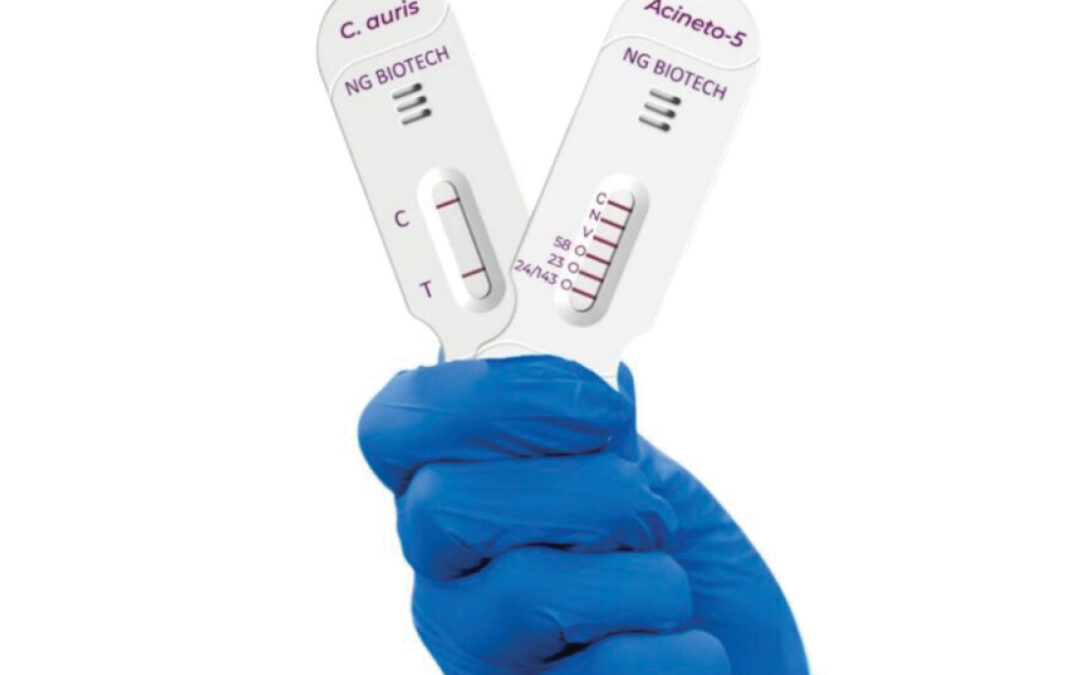
In both cases, the diagnostic tests report results back within 15 minutes

But anti-regulatory stance taken by Trump administration could serve as a potential obstacle in the effort

TruVerus is designed to perform a slew of clinical laboratory tests on as few as eight drops of blood

The two assays identify threats prior to prostate cancer being diagnosed and after it has undergone extensive treatment, respectively