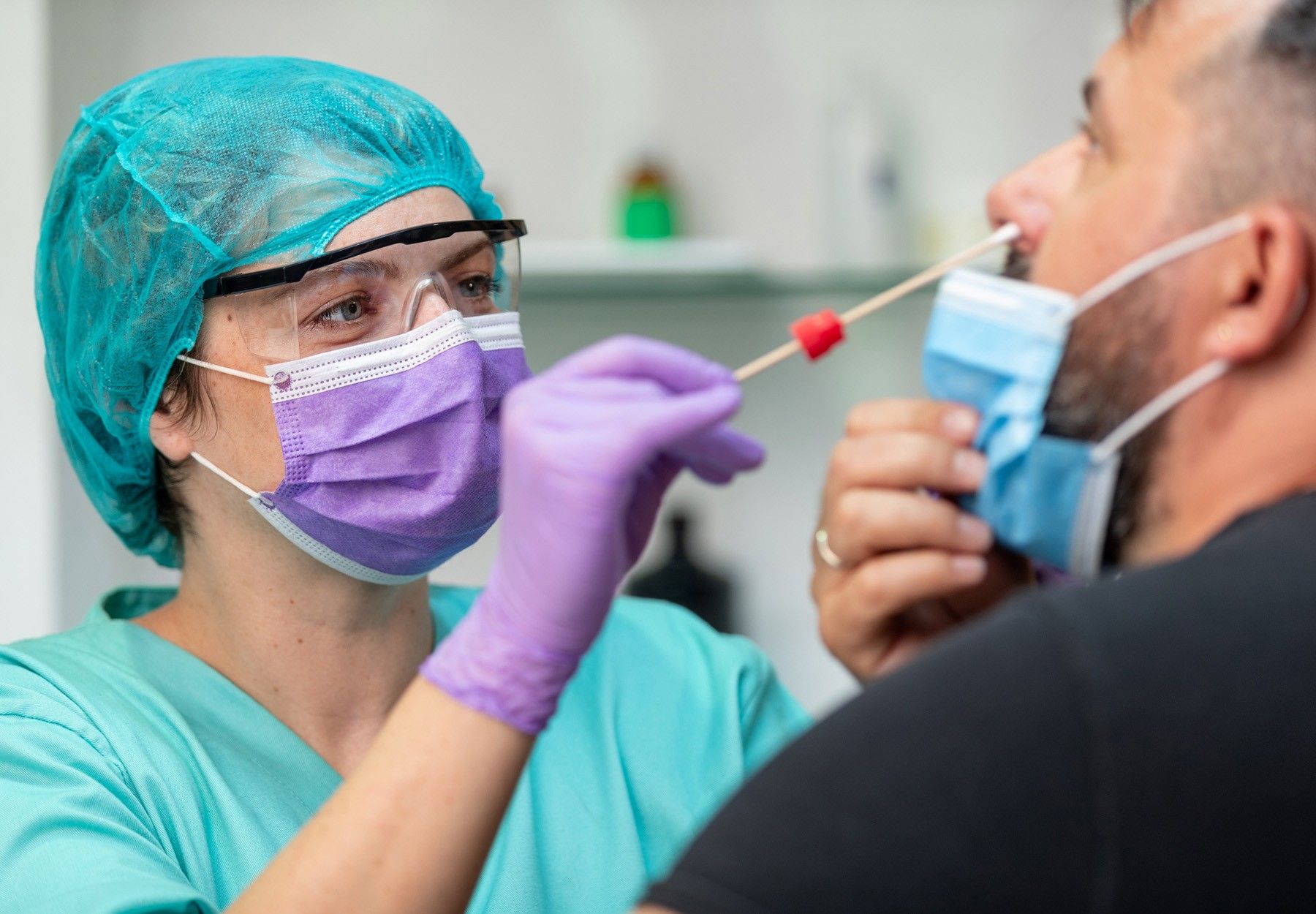
Recent Recall a Reminder to Check Which COVID-19 Tests Your Lab Can Use
Celltrion’s recall was labeled by the FDA as Class I, due to the kits possibly being distributed to non-CLIA certified users.

Celltrion’s recall was labeled by the FDA as Class I, due to the kits possibly being distributed to non-CLIA certified users.

The changes affect the Office of Product Evaluation and Quality’s Office of In Vitro Diagnostics and Radiological Health.

Agency warns the public to be on the lookout as counterfeit at-home OTC COVID-19 diagnostic tests appear on the US market.

In this month’s Business Roundup, we cover key happenings including Medicare coverage and reimbursement, as well as the US surpassing Europe as medtech’s market of choice.
InspectIR COVID-19 device detects the virus from breath samples in less than three minutes, offering a new screening option.