
New Bill Would Guarantee Medicare Coverage of Newly Approved Breakthrough Devices
Congress brings back controversial Trump plan scrapped by the Biden administration.

Congress brings back controversial Trump plan scrapped by the Biden administration.

The bill is designed to provide Medicare patients faster access to innovative technology approved by the FDA.

Although the law officially took effect on March 29, the FDA says it isn’t planning to enforce the new rules until October 1.

COVID-19 test makers will either have to get full marketing approval, or take the product off the market.
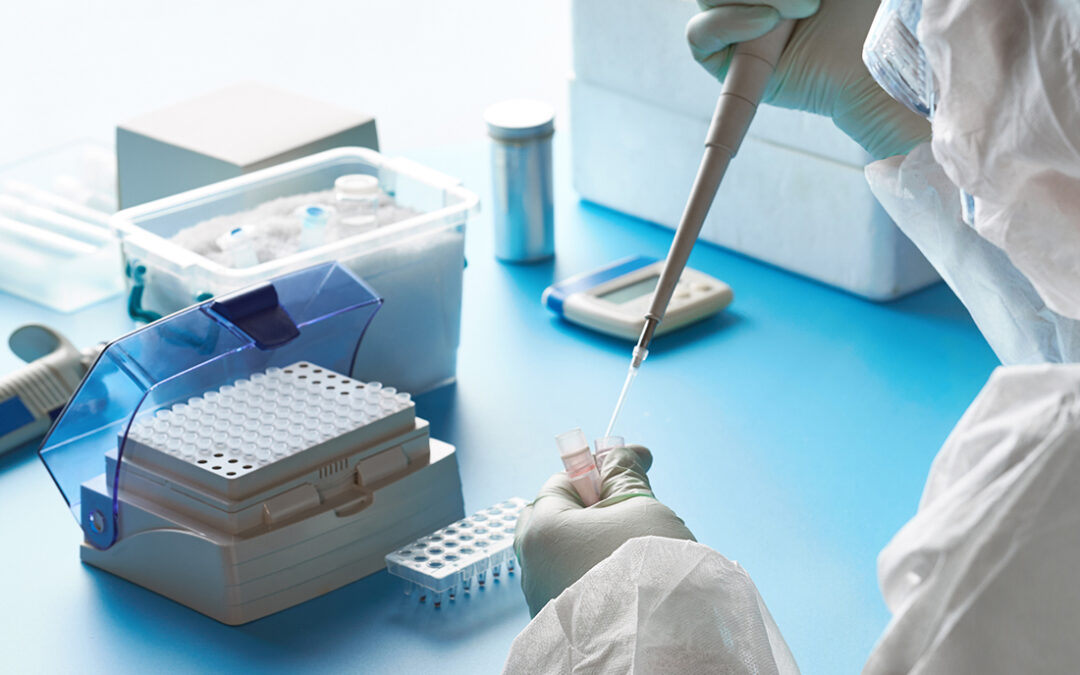
On March 27, the agency released a new guidance document to help test developers make the transition to full approval.