Keep Informed on Legal and Compliance Developments Affecting Diagnostic Labs
Best practices and expert insight to help lab professionals comply with regulations and identify strategic trends in business and technology
Already a Subscriber? Log in Here
Lab Industry Advisor Subscription
Struggling with Staying Informed? In the fast-paced world of diagnostic medicine, staying updated with the latest legal, compliance, regulatory, and industry insights is crucial. Yet, finding reliable and comprehensive information can be overwhelming and time-consuming.
Lab Industry Advisor to the Rescue! Lab Industry Advisor provides comprehensive, up-to-date insights and analyses tailored to your needs. With expert commentary and actionable advice, you’ll stay ahead of the curve. No more sifting through endless sources—everything you need is right here!
Start your free trial today and see how Lab Industry Advisor can transform your approach. Get instant access to invaluable resources designed to keep you informed and competitive. Explore detailed reports, in-depth articles, and exclusive industry insights that help you make better decisions faster. Choose from three subscription levels tailored to fit your needs: Essential, Premium, and Elite.






Additional Business Intelligence Offerings
Lab Industry Advisor Subscriptions Include

Analysis and insight on key compliance, legal, and business developments affecting laboratories
- Updates on changes to regulations
- Healthcare-related laws
- FDA approvals
- Business deals in the diagnostics space, and more
Latest Articles
LDTs Rule Update: More Support for Legal Challenges Against FDA
Key clinical laboratory organizations file amicus brief, reiterating that the FDA is overstepping its authority, and its new rule would cause “irreparable patient harm”
Medical Laboratory Personnel Shortage Relief Act Aims to Dent the Staffing Crisis
How the 2024 proposal will fare on Capitol Hill in 2025 remains to be seen, though
Image-Based AI Touted as a New Weapon Against Rare Cancers
Studies by Paige demonstrate the greater accuracy that artificial intelligence potentially offers for pathologists
Are We in a ‘Quademic’? COVID-19, Flu, RSV, and Norovirus Infections Surge
The pandemic may have set the stage for a jump in infections from these four nemeses
Bringing Clinical Mass Spectrometry into Routine Medical Laboratory Testing
Roche experts discuss the company’s recently launched automated solution and how it will bring mass spec to more patients
Diagnostics Pipeline: Roche’s New Cobas Liat Panel Can Test for Multiple STIs Simultaneously
The PCR-driven test provides results in about 20 minutes in non-laboratory settings
Dx Deals: BD Laboratory Business Is Up for Grabs in Bold Move
Becton Dickinson divisions responsible for $3.4 billion in annual revenue are on the block
Updates to CLIA Personnel Requirements May Lengthen Hiring Times
Changes ‘have had a significant impact on the recruitment process for laboratory directors and technical supervisors,’ recruiter says
Clinical Lab Efficiency Gains May Be Best Achieved Through a Tailored Approach
Optimizing efficiency unfortunately is not straightforward in medical laboratories
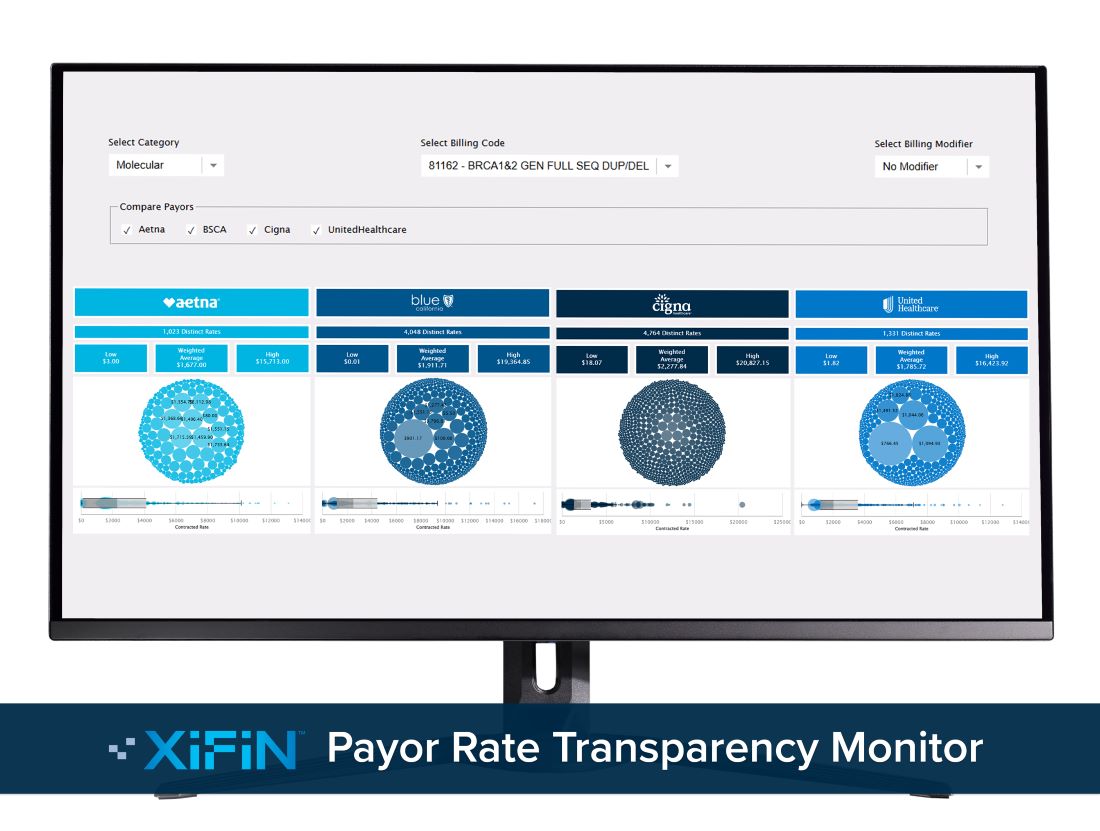
XiFin’s Payor Rate Transparency Monitor Helps Clinical Labs Compare In-Network Rates
The free online tool can set a foundation for renewed reimbursement strategies and contract negotiations with insurance carriers
Recent Decision in FCA Case Has Key Implications for Labs
The decision deepens a split among circuit courts regarding how to determine liability for false claims